Urgent Recall Issued For Specific Ozempic Pens Batch
The Malta Medicines Authority has issued a warning about a possible falsified pack of the diabetes medicine Ozempic® 1mg solution for injection in pre-filled pen.
The alert was raised by the medicine’s manufacturer, Novo Nordisk®, after a single pack from batch number PP5N682::RN287 was flagged as suspicious.
As a precaution, a patient-level recall is being carried out for this batch, which has an expiry date of February 2027.
No other batches are affected.
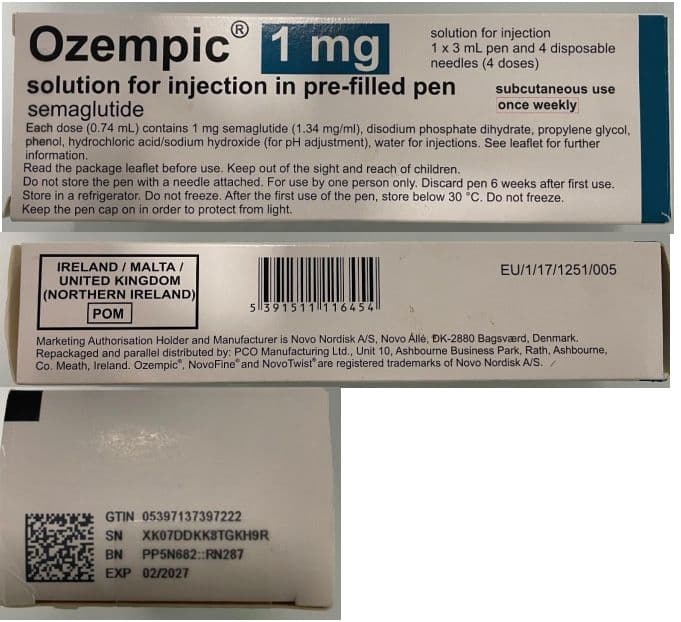
People who have Ozempic® from this batch should stop using it immediately.
They are advised to check the batch number, contact their pharmacy for verification, and return the pack to the pharmacy where it was purchased.
Patients should also speak to their pharmacist or doctor to arrange continued treatment.
The Superintendent of Public Health, together with the Malta Medicines Authority, is working closely with relevant partners to protect public health. Anyone with concerns should seek advice from a medical professional.
#MaltaDaily