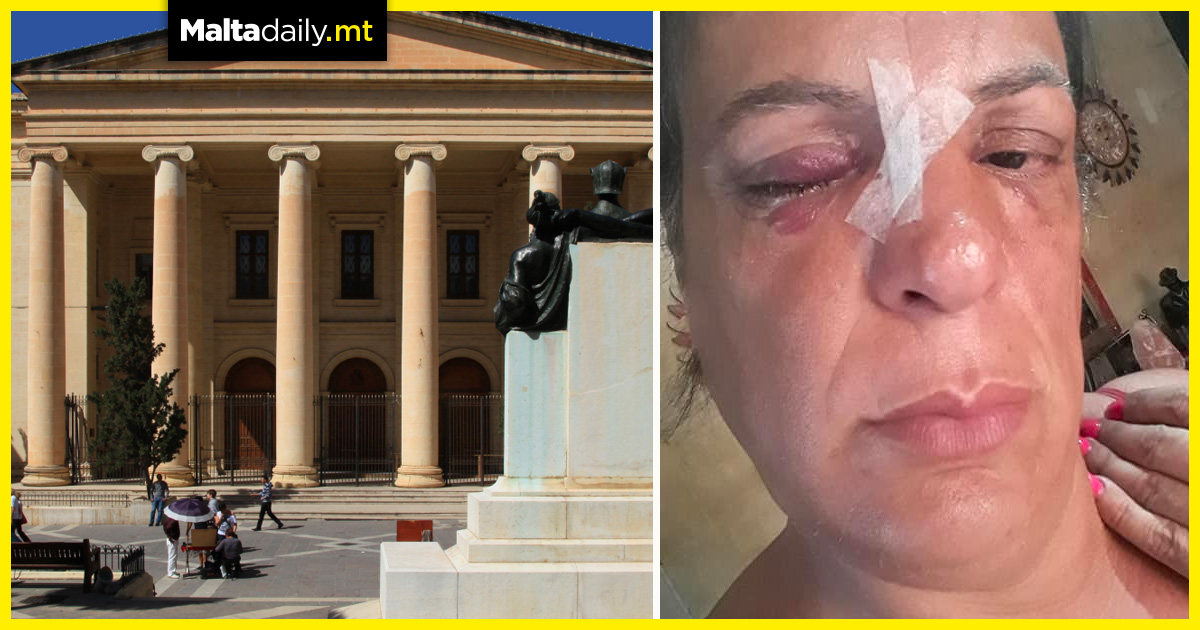
With the pharmaceutical giant Pfizer submitting research to the US Food and Drug Administration on the effectiveness of its COVID jab, it has been suggested that the shots for children may not be available until November. The company stated on Tuesday that health regulators had been provided with data from a. Recent study of its vaccine in children between 5 to 11 years of age. Officials previously stated that they would file an application with the FDA to authorise use in coming weeks.
Evidence will be reviewed and consulted with advisory committees once the company files its application. This will determine if the shots are safe and effective for recommended use. Depending on how quickly the FDA acts, jabs might be available earlier in November. Pfizer and its partner BioNTech say that they expect to request emergency use authorisation of their vaccine in the age group in the coming weeks. The companies also plan to submit data to the European Medicines Agency and other regulators.
Two-shots of the Pfizer vaccine is currently available for those 12 and older. Around 100 million people in the US have been fully vaccinated with it, according to the CDC. Pfizer tested a lower dose of the shots in children, with the drug maker saying last week that the vaccine developed corona-virus fighting antibody levels in kids which were just as strong as those found in teenagers. Moderna is also currently studying its shots in elementary school aged children.
#MaltaDaily
Photo Source: Wall Street Journal